In a recent development in the multidistrict litigation (MDL) surrounding DuPuy Pinnacle hip products, Johnson & Johnson has agreed to pay $1 billion to resolve the majority of pending claims. Claimants have filed about 10,000 DuPuy Pinnacle hip lawsuits in the last several years over allegations of personal injury caused by the device.
DePuy Pinnacle Hip Lawsuits
Due to the thousands of Pinnacle hip claims filed, the cases were consolidated in 2011 into MDL 3:11-md-02244. The MDL is being handled by the U.S. District Court in the Northern District of Texas.
Several trials have occurred as a result of the issues with Pinnacle hip devices, and several settlements have been reached. Most recently, juries in this matter approved a $1 billion settlement which should resolve about 95 percent of the remaining Pinnacle hip lawsuits.
This move is only the most recent development in this lengthy and ongoing process. Previous decisions in the case included:
- DePuy tried and lost three Pinnacle hip cases and was ordered to pay more than $1.7 billion in damages to those victims alone.
- In February 2018, Johnson & Johnson paid $120 million to settle 46 suits brought by state Attorney General’s over the disingenuous claims of the Pinnacle hip’s durability.
- Johnson & Johnson previously paid $400 million to resolve law firm inventories of Pinnacle hip cases.
Pinnacle Hip Injury Claims
The victims who have filed lawsuits related to Pinnacle hip devices allege that the implanted devices have caused them:
- Debilitating pain
- Limited mobility
- Metal poisoning (metallosis)
- Device failure that prompts corrective surgeries
- Complete failure of the device requiring removal
Numerous claims have also related to the belief that Johnson & Johnson and DePuy failed to properly inform patients and healthcare providers about the risks of the Pinnacle hip devices.
More Hip Replacement Lawsuits in the News
These large settlements related to the Pinnacle hip medical devices came close on the heels of litigation surrounding DePuy’s other embattled hip replacement product, DePuy Articular Surface Replacement (ASR). The company resolved 93,000 claims with a settlement of $2.5 billion in 2013. DePuy also faced regulatory action from the U.S. Food and Drug Administration (FDA). Pinnacle hip products were not involved in the regulatory action.
In recent years, other hip device makers have also been the subject of personal injury lawsuits for similar complaints, including:
- Zimmer
- Stryker
- Biomet
- Wright
- Smith & Nephew
Like Johnson & Johnson and DePuy, these manufacturers are alleged to also have mislead patients and healthcare providers, and marketed products that caused injury. Many of the patients filing claims have also indicated metal poisoning, bone damage, or complete device failure.
No Pinnacle Hip Recall Despite Claims
Despite the three lost jury trials, tens of thousands of claims of injury, and billions of dollars paid in legal action settlements, DuPuy Pinnacle hip products have not been recalled. Johnson & Johnson continues to insist the devices serve a greater benefit than the risk they pose to patients. Consequently, the products are still being used.
The FDA has not taken any steps to force a recall of the Pinnacle hip device despite the 1,300 reports of adverse events the agency has tallied. Alarmingly, the majority of FDA recalls are voluntary, meaning that the manufacturer initiates the recall. It defies explanation why the manufacturer still confidently defends this product. Furthermore, it is startling that the FDA will not take action in the midst of obvious negligence.
About Metal on Metal Hip Implants
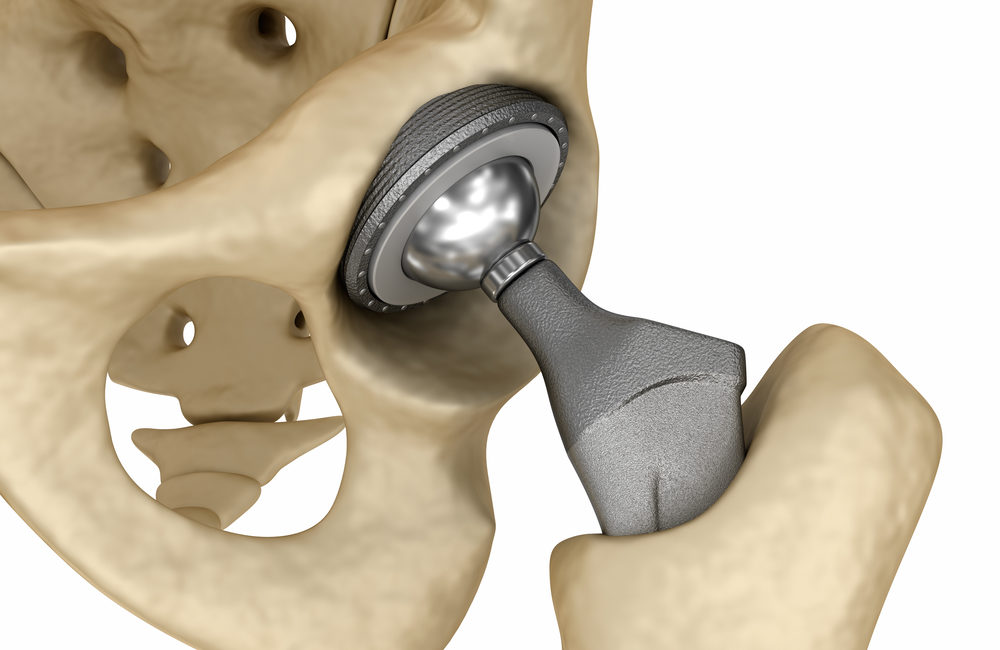
DePuy Pinnacle hip implants have been associated with adverse effects largely related to their metal-on-metal design. Hip implants in general are medical devices capable of restoring mobility and relieving pain in patients who suffer from arthritis, bone disease, or injuries. The effectiveness of the Pinnacle hip system is questionable.
Metal-on-metal hip implants are reproductions of the ball-and-socket joint found in the hip. The devices can be made of several different materials, including:
- Metal-on-Polyethylene – The ball is metal, and the socket is plastic or plastic-lined.
- Ceramic-on-Polyethylene – The ball is ceramic, and the socket is plastic or plastic-lined.
- Ceramic-on-Ceramic – The ball is ceramic, and the socket is ceramic-lined.
- Ceramic-on-Metal – The ball is ceramic, and the socket is metal-lined.
- Metal-on-Metal – Both the ball and socket are metal or metal-lined.
Metal-on-metal hip implants lost FDA approval in 2016. Both DuPuy Pinnacle hip implants and their ASR implants feature metal-on-metal design. Between 2000 and 2015, about two million patients received a Pinnacle hip implant before the metal-on-metal device lost FDA approval.
Injuries Caused by Pinnacle Hip Implants
A hip replacement is major surgery. As a result, there are quite serious risks that apply to all surgeries, including blood clots, stroke, and heart attack. Hip replacement surgeries in general put patients at risk of joint infection, bone loss, a difference in leg lengths, and hip dislocation. All hip implants pose a risk of device failure, but users of the DuPuy Pinnacle hip claim that the device breaks down and fails too easily and too soon after implantation.
Signs that a hip replacement is failing and may require corrective or revision surgery include:
- Pain in the hip, leg, or groin
- Swelling near the joint
- A sudden limp or change in walking ability
- Noise from the hip joint – popping, grinding, squeaking, and clicking
All hip implants also carry the risk of wear of the component materials. The wear of metal on metal inside the body puts the patient at risk of:
- Systemic reactions to metal particles or ions
- Heavy metal poisoning (metallosis)
Surgeons do their best to minimize the sliding and grinding of an artificial joint. However, there is no way to completely prevent or contain breakdown of materials in an artificial joint once it is in use. The metals in the Pinnacle hip implant are cobalt and chromium – two metals no one wants in their bloodstream.
Metal Ions in the Bloodstream
Every patient will react to the presence of metal ions in the blood in a unique way. For some patients, the irritation of loose metal shards will only affect the localized area around the implant and prompt earlier replacement and revision surgeries. For those unlucky enough to suffer severe and systemic reactions to the metal debris loose in the body or bloodstream, the risks include:
- Skin rashes
- Cardiomyopathy (thickening of the heart muscle)
- Hearing or vision impairments
- Depression
- Cognitive impairment
- Impaired kidney function
- Thyroid dysfunction
Among the two million patients who have metal-on-metal hip implants, those at highest risk for severe systemic reactions are:
- Patients who have had both hips replaced (bilateral replacement)
- Female patients
- Patients who take high doses of corticosteroids
- Overweight patients
- Patients with preexisting kidney problems
- Individuals with suppressed immune systems
- Patients with sensitivities to heavy metals
- Athletic patients, or patients with a high level of physical activity
Learn More about Pinnacle Hip Implant Lawsuits
While a recall can certainly spark consumer attention, a recall is not necessary in order to take legal action. Patients who have been harmed by DuPuy hip products certainly have legal recourse. Experiencing hip pain, noise, sudden difficulty walking, a weakness of the hip joint, or any sign of heavy metal poisoning is certainly cause for concern. Consult with your orthopedic surgeon if you experience these symptoms. Your hip implant needs to be evaluated immediately.
It is also recommended that patients report any adverse events associated with medical devices to the FDA. This aids in their ongoing effort to track the impact of this medical device.
Finally, call Drug and Device Watch if you have questions or concerns about your use of a hip implant. Our team of medical device attorneys have helped numerous clients like you who trusted the Pinnacle hip implant but instead have suffered negatively.
If you have suffered months or years of pain, infection, corrective surgery, or device failure, contact us today. There may still be time to file your claim. Find out how to get started by submitting our contact form or by calling 1-888-458-6825 today.
Sources:
- https://www.neuralit.com/news/2019-may-08/pinnacle-hip-lawsuit-jj-agrees-pay-1-billion
- https://www.fda.gov/medical-devices/metal-metal-hip-implants/concerns-about-metal-metal-hip-implants
- https://www.depuysynthes.com/hcp/hip/products/qs/PINNACLE-Hip-Solutions
- http://www.txnd.uscourts.gov/mdl-311-md-02244
- https://www.latimes.com/business/la-fi-johnson-johnson-settle-pinnacle-metal-hip-implant-20190507-story.html