Otsuka Pharmaceuticals and Bristol-Myers Squibb
In 2016, the FDA discovered 184 cases that linked Abilify to compulsive behaviors. Patients taking the medication experienced uncontrollable compulsions in the areas of:
- Gambling
- Shopping
- Eating
- Sexual behavior
- Impulse-control
Patients may also experience addiction to these compulsions, and may be unable to stop themselves even if they are causing themselves or others injuries.
Roche/Genentech
Actemra is a drug used to treat rheumatoid arthritis. Though there is no such warning on the label, Actemra is known to increase the risk of:
- Stroke
- Heart failure
- Pancreatitis
- Lung disease
- Gastrointestinal perforations
Actemra has been widely marketed as not having these risks. An investigation shows that Actemra carries a higher risk of these adverse events than other similar RA drugs.

Takeda, Inc.
Actos is a drug used to treat Type 2 Diabetes. In 2011, it was removed from European markets after studies showed that patients taking Actos developed bladder cancer at an increased rate.
The drug is still available in the U.S. The FDA now requires warnings about the increased risk of bladder cancer be attached to packaging.
GlaxoSmithKline
Studies show that Avandia may cause congestive heart failure or myocardial ischemia. In 2011, the FDA released a safety notice about the increased risk of congestive heart failure and heart attack. The FDA also requires a black box label be applied to packaging.

Bayer Corporation
In 2001, Bayer announced that it was stopping production of the cholesterol medication Baycol due to a significant number of adverse event reports. Numerous people have reported serious side effects and at least 100 people have died after taking the medication.
Baycol can cause an illness called rhabdomyolysis, which cause pain and weakness in the back, calves or entire body. Patients may also experience fever, nausea and vomiting and dark urine.

Daiichi Sankyo
Thousands of claims were filed against Daiichi Sankyo after patients developed sprue-like enteropathy and other types of gastrointestinal illness while taking the medication. Patients argue that the company did not warn them about the risks of taking Benicar.
Sprue-like enteropathy is an illness that causes chronic diarrhea, nausea, weight loss and malnutrition.
Actavis Totowa, LLC
Digitek is a drug used to treat abnormal heart rhythms. In 2008, the drug was recalled due to the possibility that pills contained double the amount of the active ingredient. The result was patients experiencing digitalis toxicity.
Digitalis toxicity causes severe symptoms including:
- Dizziness
- Nausea and vomiting
- Low blood pressure
- cardiac instability
- Bradycardia
Numerous patients have reported these adverse events while taking Digitek.
Janssen Pharmaceuticals
Numerous Elmiron lawsuits have been filed after patients experienced maculopathy – an eye disorder that damages the retina and can cause blindness.
Elmiron lawsuits claim that Janssen Pharmaceuticals did not properly warn patients about the risk of vision problems or permanent injury. Research clearly links the ingredients in Elmiron to ocular toxicity and permanent and irreversible damage to the eyes.
AstraZeneca
Diabetes medication Farxiga has been linked to Fournier’s Gangrene, a deadly flesh-eating bacteria that affects the genitalia and perineum. Fournier’s Gangrene occurs when body tissue dies due to a bacterial infection or loss of blood supply.
Over a 30 year period, there were only 19 cases of Fournier’s Gangrene reported. However, in the 15 years after market approval of Farxiga, there were 55 cases linked to the drug.
Bayer, Bracco, Guerbet, Mallinckrodt
Between 2008 and 2015, over 1,000 lawsuits were filed against manufacturers of Gadolinium-Based Contrast Agents (GBCA). Contrast is the drug injected into a patient before an MRI or MRA.
Reports show that certain GBCA’s may cause Gadolinium toxicity leading to:
- Nephrogenic Systemic Fibrosis
- Skin hardening
- Joint contracture
- Chronic pain
- Loss of mobility
- Gadolinium Deposition Disease
- Pins and needles
- Intense pain in joints, tendons, ligaments, and bones
- Mental confusion
- Numbness or tingling
- Persistent headache
- Kidney damage
- Rubbery skin
Fresenius
GranuFlo is an acid concentrate that is used during dialysis treatment. Since it’s release, GranuFlo has been linked to adverse events, including heart attacks in patients.
Patients receiving dialysis may be unable to process and filter acid buildup caused by GranuFlo. The result is metabolic alkalosis, which can cause heart attacks.
A number of lawsuits have been filed against Fresenius.

American Regent
Injectafer (ferric carboxymaltose) is an iron replacement medication prescribed to patients for intravenous administration. Injectafer lawsuits claim that patients and the medical community were not adequately warned of the risks of Injectafer, including adverse events such as:
- Hypophospatemia (HPP) – results in neuromuscular, cardiovascular and hematologic dysfunction
- High blood pressure
- Cardiac arrest
- Respiratory failure
- Seizures
- Coma
- Death

Johnson & Johnson
Clinical trials show that Invokana – a Type 2 diabetes medication – doubles the likelihood that a patient will require a foot or leg amputation. Patients taking Invokana should be mindful of the following symptoms:
- Soreness
- Ulcers
- Tenderness
Numerous lawsuits claim that J&J failed to warn doctors and patients about the increased risk of ketoacidosis, kidney failure and amputations.

Sanofi-Aventis
Ketek, or generic telithromycin, is an antibiotic used to treat chronic bronchitis and respiratory infections. Beginning in 2001, the FDA released safety information about Ketek and possible adverse events while taking it. Adverse events included:
- Liver injuries
- Liver toxicity
- Blurred vision
Since 2006, at least three patients have reported severe liver toxicity. One patient has died.
Pfizer
Lipitor is a statin, or a cholesterol-lowering medication. Many lawsuits have been filed against Pfizer claiming that patients taking Lipitor experienced severe reactions and adverse events.
In addition to common side effects, studies show that Lipitor may increase the risk of women developing Type 2 diabetes by 48%.

GlaxoSmithKline
Paxil (paroxetine hydrochloride) is a selective serotonin re-uptake inhibitor (SSRI) used to treat anxiety, depression and related disorders. Paxil has been linked to serious adverse events, including bouts of extremely violent behavior and suicidal ideation. Paxil has also been linked to birth defects.
The FDA has issued warnings about adverse events and has requested manufacturers include stronger warnings on their labels.
Several lawsuits have been filed by victims who have experienced birth defects, violent behavior or the loss of a loved one to suicide.
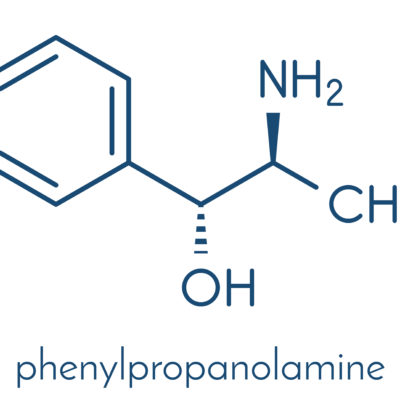
Various
Beginning in 2000, the FDA began attempts to remove all products containing PPA from the market. PPA is an ingredient in many prescription and over-the-counter (OTC) medications. Most often, these medications are used for cold and cough, decongestants and weight loss.
Scientists have found a link between PPA and hemorrhagic strokes, primarily in women. The FDA urges consumers to avoid PPA if possible.
Some common products that contain PPA include:
- Alka Seltzer cold medicines
- BC Allergy Sinus Powder
- Comtrex
- Coricidin
- Dexatrim
- Dimetapp
- Robitussin CF
- Triaminic
Janssen Pharmaceuticals
Propulsid is an effective drug to treat heartburn and acid reflux. Unfortunately, shortly after approval, patients began to report serious adverse events, including heart problems. There is also evidence that Propulsid may be dangerous to children.
There have been numerous lawsuits filed against Janssen and Johnson & Johnson, the parent company. In 2004, J&J paid around $90 million to settle lawsuits by 4,000 patients who experienced heart problems while taking Propulsid.

Fresenius Kabi
Protamine sulfate is an antidote used to treat patients who overdose on heparin. Recently, a number of patients have reported experiencing severe cardiac complications after being administered Protamine. Some patients have died as a result.

Genetech, Inc.
In 2009, the FDA became aware of at least four cases of multifocal leukoencephalopathy (PML) among patients taking Raptiva. PML is a severe brain infection that can be deadly. An estimated 80% of people who develop PML die in six months or less.
Raptiva is also linked to adverse events including:
- Bacterial sepsis
- Invasive fungal disease
- Viral meningitis

ANI Pharmaceuticals, Inc.
Reglan (metoclopramide) is used to treat a variety of gastrointestinal illnesses. Unfortunately, this popular drug has been linked to serious adverse events. Reglan is linked to Tardive Dyskinesia, a central nervous system (CNS) disorder. Tardive Dyskinesia causes involuntary movements and twitching of the limbs, face and tongue.

Warner-Lambert Co.
Rezulin is a Type 2 diabetes medication used to help control insulin resistance. In 1997, patients began to report instances of liver failure after taking Rezulin. In 2000, the FDA removed Rezulin from the market. Since approval in 1997, it is thought that as many as 400 people may have died due to liver failure and heart problems caused by the drug.

Bristol-Myers Squibb
Serzone is an antidepressant medication approved by the FDA in 1994. There are common side effects to Serzone, but also the possiblity of serious or life-threatening hepatic failure.
In 2001, the FDA ordered a “black box warning” on the label. In 2003, Serzone was pulled from the European market after 25 reports of liver failure. Eighteen people died.
Serzone was pulled from the U.S. market in 2004. Bristol-Myers Squibb cites poor sales as the reason, not the potentially life-threatening adverse events experienced by numerous patients.

Johnson & Johnson
Johnson & Johnson talcum powder products have been used for feminine hygiene for many years. Sadly, many women who have used talcum powder have developed mesothelioma or ovarian cancer.
Scientists believe there is a link between talcum powder products and these dangerous cancers. Several studies show that women who used J&J talcum powder were more likely to develop ovarian cancer than other women. Research also shows that J&J knew their products contained asbestos, but did not publicly disclose this fact.
There are currently more than 19,000 lawsuits on record.

Sanofi-Aventis
Chemotherapy drug Taxotere has been linked to permanent hair loss in women taking the drug. Numerous reports of women developing alopecia after taking Taxotere have resulted in the FDA including a warning on the drug.
Numerous lawsuits have been filed and consolidated in federal court. Patients report that had they known about the risk of permanent hair loss, they would have chosen a different chemotherapy drug.

Bristol-Myers Squibb
Tequin (gatifloxacin) is an antibiotic drug used to treat patients with bacterial sinus, lung or urinary tract infections (UTIs). Unfortunately, Tequin has been linked to several potentially serious side effects, including:
- Renal failure
- Hypoglycemia (low blood sugar)
- Hyperglycemia (high blood sugar).
In 2006, Bristol-Myers Squibb stopped manufacturing and selling Tequin. Side effects have been reported in many parts of the world.
Various
In 2015, the FDA issued a Safety Announcement about certain testosterone therapies. In the announcement, the FDA began requiring manufacturers to change their labels to clarify approved uses. The FDA also required labels to include a warning about the increased risk of heart attack and stroke. Studies suggest that testosterone replacement therapy may increase the risk of heart attack by 30%.
Thousands of lawsuits have been filed against manufacturers. Lawsuits claim that patients taking testosterone experienced heart attack or stroke. Lawsuits also claim that patients were not adequately warned of the risks before taking these medications.

Janssen Pharmaceuticals
The FDA warns consumers that Topamax causes and increased risk of birth defects if taken during pregnancy. The popular epilepsy and migraine medication is linked to a 21-times higher risk of cleft palate deformities. It is also linked to other birth defects including:
- Genital birth defects
- Spina bifida
- Hypospadias
- Craniofacial defects
- Neural tube defects
- Heart defects
- Lung defects

Bayer HealthCare
Trasylol is a drug that was used to control bleeding. It was most often used in patients who needed Coronary Artery Bypass Grafting surgery. Research shows that Trasylol is linked to an increased risk of:
- Heart attack
- Stroke
- Kidney failure
- Death
Trasylol was taken off the market in 2007, but not before it was linked to potentially 22,000 deaths.
Various
Acetaminophen, the primary ingredient in Tylenol, has been the subject of various lawsuit due to possible liver toxicity. U.S. Poison Control centers report more calls related to acetaminophen each year than any other drug. It is estimated that around 100,000 calls are made each year regarding adverse events from acetaminophen.
It is also estimated that more than 50,000 emergency room visits each year result from adverse events of acetaminophen, including:
- Nausea and vomiting
- Upper abdominal pain
- Yellow skin
- Dark urine
- Diarrhea
These are all possible symptoms of liver damage, which is a known side effect of acetaminophen.

Takeda Pharmaceuticals
Uloric is a xanthine oxidase inhibitor used to manage chronic hyperuricemia (excessive uric acid) in patients who have gout. Uloric was approved by the FDA in 2009. In 2019, the FDA mandated that Takeda place warning labels on the drug warning consumers about the risk of cardiovascular problems.
Research shows that patients who take Uloric have a significantly increased risk of cardiovascular problems and death compared to patients who take alternative drugs.

Tesaro, Inc.
Varubi is a drug used to treat chemotherapy-induced nausea and vomiting. It is available in an oral tablet and intravenous formula. In 2018, Tesaro announced updates to the drug’s label after receiving adverse event reports about patients suffering from anaphylaxis, anaphylactic shock and other hypersensitivity reactions. These reactions occur within minutes of taking the drug, and result in:
- Hives
- Respiratory distress
- Swelling
- Itching
- Chest pain
- Low blood pressure
Numerous lawsuits were filed against Tesaro claiming that the company provided misleading and materially false information about the danger of allergic reactions.

Merck & Co.
Vioxx is a selective cox-2 inhibitor that is used to manage pain associated with arthritis. It is also used to treat minor pain related to headaches and menstrual pain. It is available in tablet and liquid form. There are known minor side effects related to Vioxx, but in 2002, the FDA became aware of a number of cases of nonbacterial meningitis among patients taking the drug. There were also numerous reports of patients suffering sudden cardiovascular events, including stroke and heart attack.
Vioxx was removed from the market in 2004. Lawsuits filed against Merck claim that patients suffered heart attack, stroke and even death. In 2007, Merck settled a number of these cases for $4.85 billion.

Genentech, Inc.
Xolair is a “humanized antibody” – a drug that is derived from a non-human source. This drug was derived from the ovaries of the Chinese Hamster. The intravenous drug is used to treat moderate or severe asthma in adults and children older than 12. Xolair has been linked to an increased risk of cerebrovascular and cardiovascular problems. Patients report serious allergic reactions, including:
- Severe skin rash
- Drop in blood pressure
- Join and limb pain
- Anaphylaxis
In 2019, Xolair was recalled due to these potential risks and the possibility of serious heart and brain problems.
Bayer HealthCare Pharmaceutical
Yaz, Yasmin and Ocella are birth control pills that contain drospirenone. Drospirenone is a synthetic version of progestin, a hormone that works with estrogen to prevent pregnancy. Drospirenone has been linked to serious adverse events and injuries in women taking the drug. One study shows that women taking drospirenone may be at a 200% increased risk of serious injuries, such as:
- Heart attack
- Stroke
- Pulmonary embolism
- Deep vein thrombosis (DVT)
- Cardiac arrhythmia
- Kidney failure
- Sudden death
Sanofi
The U.S. Food and Drug Administration (FDA) confirms laboratory tests that ranitidine products contain higher-than-acceptable levels of N-nitrosodimethylamine (NDMA). The probable human carcinogen is known to increase the risk of certain cancers.
Sanofi is the manufacturer of brand name Zantac, but several manufacturers of generic ranitidine have also pulled their products from the market. These include:
- Sandoz
- Apotex
- Perrigo
- Dr. Reddy’s Laboratories
- Lannett Company Inc.
- Novitium Pharma LLC
- Aurobindo Pharma USA
- American Health Packaging
- Denton Pharma, Inc. dba Northwind Pharmaceuticals
- Amneal
- Appco Pharma LLC
- Glenmark Pharmaceuticals, Inc.
- Generic store brands:
- CVS
- Rite-Aid
- Walgreens
- Wal-Mart

Novartis Pharmaceuticals Corporation
In 2007, the FDA sent a notice to healthcare providers and patients that Novartis was discontinuing Zelnorm. Zelnorm was used to treat patients with irritable bowel syndrome (IBS) accompanied by constipation. The FDA became aware of clinical data showing that an excessive number of patients taking Zelnorm experienced cardiovascular events, such as:
- Heart attack
- Stroke
- Angina
As a result, Zelnorm was pulled from the market.
Pfizer, Inc.
Zithromax, or Azithromycin, is an antibiotic often used to treat bacterial infections. The antibiotic interferes the bacteria reproducing. Zithromax is considered highly effective, but has also been linked to some serious adverse events. Research shows that patients taking Zithromax may be 1.5 times more likely to experience sudden cardiac problems, including sudden death. The risk is greatest among patients with prior heart problems or surgeries.

GlaxoSmithKline
Zofran is a widely prescribed drug used to treat nausea and vomiting. It is often prescribed to patients with severe gastrointestinal illnesses, as well as patients undergoing chemotherapy or radiation. It is also often used “off label” to relieve the symptoms of morning sickness during pregnancy. Numerous lawsuits have been filed against GSK, the manufacturer, claiming that taking Zofran while pregnant led to birth defects.
Lawsuits further claim that GSK knew there was a risk of birth defects, but did not warn patients, healthcare providers or regulators.
Call us at (888) 458-6825 or fill out our contact form to discuss your legal options.
The consultation is free and confidential.