Bayer Essure
The Essure birth control implant was approved in 2002. From that time until December 2019, the U.S. Food and Drug Administration (FDA) received more than 47,000 adverse event reports from patients.
Essure is no longer available in the U.S. but the FDA continues to collect information from women who have used the device.

Essure Birth Control Implant
Adverse events related to the Essure device include:
- Abdominal pain
- Irregular menses
- Headache
- Perforation
- Device fragments in the body
- Device migration
- Depression/anxiety
- Weight fluctuations
- Device failure
- Ectopic pregnancy
There are 57 reports of death related to Essure – some among women and some among infants.
Surgical Staplers
Surgical staplers are medical devices used to close incisions or lacerations. They have been on the market for more than 50 years and are believed to be responsible for more than 41,000 adverse events. Numerous lawsuits allege that surgical stapler malfunctions have caused serious injuries to patients. The devices believed to be responsible for these injuries include:
Covidien EEA Circular Stapler with Tri-Stapler Technology
In 2018, Covidien recalled the EEA Circular Stapler with Tri-Stapler Technology due to a malfunction that could cause the stapler to have an incorrect tissue gap. This malfunction resulted in incomplete staple formation and difficulty removing the device from the patient’s tissue.
The malfunction and tissue gap could cause tissue trauma, anastomotic leak or bleeding.

Covidien EEA Hemorrhoid and Prolapse Stapler Sets
In April 2018, Covidien recalled EEA Hemorrhoid and Prolapse Stapler Set with DST Series surgical staplers. Improper welding on the staple guide could result in improper staple formation. Patients may experience adverse events including anastomotic leak and bleeding.

Covidien GIA Surgical Stapler
In 2018 and 2019, Covidien recalled GIA surgical staplers commonly used after gastrointestinal procedures. The 2018 recall was due to a missing sled component that could result in the device not forming a staple line. The 2019 recall related to missing pin components that could affect the alignment of the stapler.
Adverse events associated with these surgical staplers include:
- Bleeding
- Peritonitis
- Anastomotic leak
- Pneumothorax
- Increased risk of infection or sepsis
Orthopedic Devices
In recent years, numerous orthopedic devices have been listed as potentially dangerous. When these devices are defective or malfunction, patients can experience adverse events that affect their health, safety, and quality of life. Some of the most concerning orthopedic devices include the following:
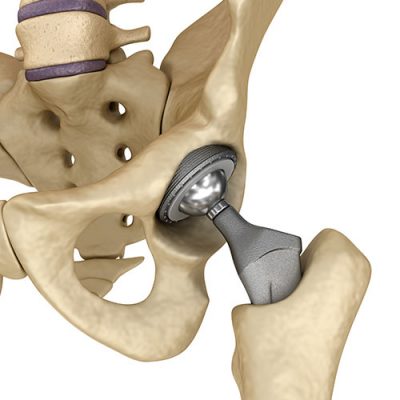
Biomet Hip Implants
Biomet metal-on-metal hip implants (M2A 38mm/Magnum) may cause metallosis, or metal poisoning. Metal particles may come loose and enter the bloodstream causing pain, damage to surrounding tissue, device failure, kidney problems, and the need for revision surgery.
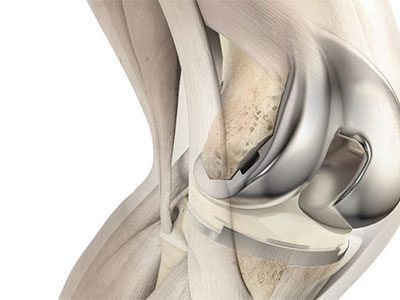
DePuy Synthes Knee Implants
The ATTUNE® Knee System is reported to have an “unusually high rate of early failure”. The implant may debond within two years, causing swelling, limited range of motion, pain, fluid build-up, or device failure. Patients may require revision or replacement surgery.

DePuy Pinnacle Hip Solutions Implants
Beginning in 2010, thousands of complaints have been filed against DePuy and Johnson & Johnson after consumers found that the orthopedic device did not last as long as the company’s advertised them to. According to complaints, the device was advertised to have a greater than 90 percent survival rate over five years. European health regulators, however, found the rate to be closer to five percent. Several thousand more complaints were filed alleging that the metal-on-metal implants caused metal poisoning.

DePuy ASR 300 Acetabular Cup System
In 2010, the ASR 300 system was recalled after a report indicated that the number of patients requiring additional hip surgery were much higher than previously reported. Patients were experiencing high rates of symptoms including loosening of the device, fractures to surrounding bone, dislocation, swelling, and severe pain.
Hernia Mesh

Sofradim Production Versatex Monofilament Mesh
In 2018, Sofradim Production recalled the Versatex Monofilament Mesh device after reports of abdominal hernia recurrence after the mesh was installed. Hernia recurrence is often a sign that surgical mesh was not successful, was defective, or has failed. Additional hernia repair surgery is often required.

Atrium Medical Corporation ProLite Mesh
Atrium Medical Corporation issued a recall of ProLite Mesh devices in 2017. The mesh was placed in packages with the incorrect product code. This could result in surgical use of the wrong product.

Atrium Medical Corporation C-QUR Edge Mesh
In 2013, Atrium Medical Corporation recalled C-QUR Edge Mesh (all shapes and sizes) due to packaging issues causing the device to be exposed to increased humidity. The humidity was found to cause the device to adhere to the lining of the packaging and handling sleeve. Adhering to the packaging could cause the coating on the mesh to peel off, compromising the device.

Ethicon Polymeric Surgical Mesh
Ethicon Polymeric Surgical Mesh was recalled in 2010 due to the potential risk of delamination. Delamination of the product could compromise sterility and increase the risk of side effects and device breakdown.

Bard Kugel Mesh
Bard Kugel Mesh products were recalled in 2006 due to the risk of the recoil ring breaking. This breakage could result in bowel perforation and/or chronic enteric fistula.
Call us at (888) 458-6825 or fill out our contact form to discuss your legal options.
The consultation is free and confidential.