The latest and final step in an escalating series of warnings and actions about surgical mesh for the treatment of pelvic organ prolapse (POP) came this week as the U.S. Food and Drug Administration (FDA) ordered the manufacturers of some mesh products to stop selling and distributing the devices immediately.
The End of Surgical Mesh for POP
There are many types of surgical mesh, but the FDA’s order refers specifically to the mesh used in transvaginal repair of pelvic organ prolapse (POP). Surgical mesh used to treat POP transvaginally has been on the FDA’s list of Class III products – those with the highest risk – since 2016.
Since that time, the FDA has required manufacturers to adhere to their most rigorous device review pathway in order to market their devices in the U.S. with the agency’s approval. The process requires manufacturers to submit premarket approval (PMA) applications to the FDA to demonstrate the safety and effectiveness of their products.
The order to immediately stop marketing this specific mesh medical device came after the FDA rejected two PMA applications from the two manufacturers still marketing transvaginal surgical mesh – Boston Scientific, and Coloplast. The order effectively announces that these surgical mesh products pose too great a risk to consumers.
In a statement issued April 16, 2019, the FDA says manufacturers have failed to provide evidence that surgical mesh is a more effective treatment for POP than surgeries that don’t use the mesh. The FDA’s rejection and subsequent removal order means these companies have little more than a week to submit a plan to the agency about how they will withdraw their devices from the medical device marketplace.
The Use of Surgical Mesh for POP
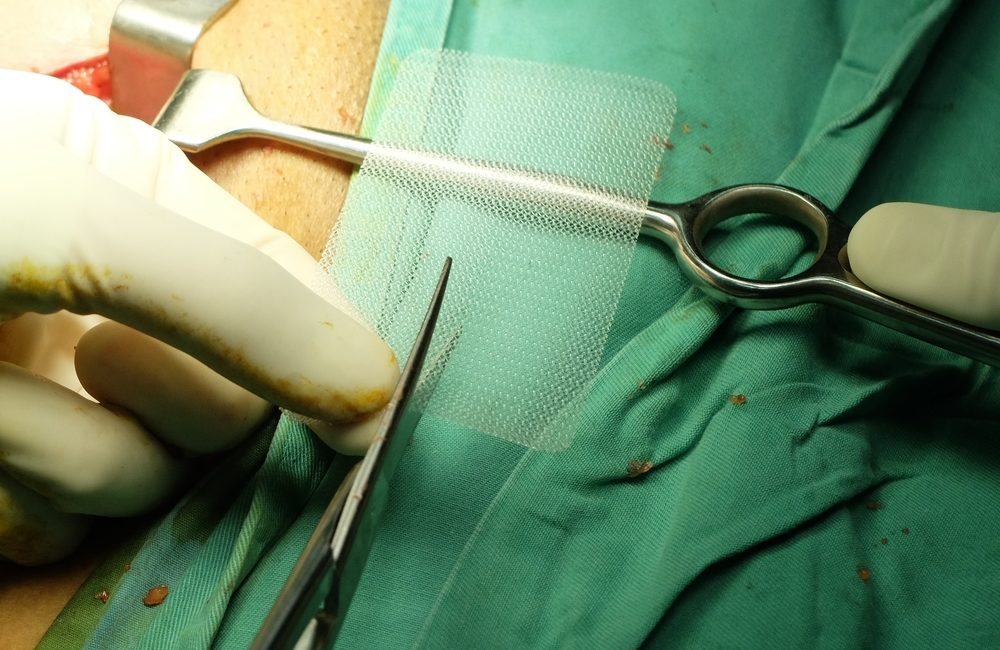
Surgical mesh is a medical device doctors use to support weakened or damaged tissue. As a device, it has many uses including:
- Hernia repair
- Repair of stress urinary incontinence (SUI)
- Repair of pelvic organ prolapse (POP)
Surgeons have used mesh devices since the 1950’s. Originally used exclusively for abdominal hernia repair, gynecological surgeons began using mesh for abdominal repair of POP in the 1970’s. By the 1990’s, surgeons developed the transvaginal procedure of repairing POP, and by 2002, manufacturers began marketing the first FDA-approved surgical mesh specifically designed to be used in transvaginal POP repair.
Only the mesh devices intended for use to treat POP transvaginally are affected by the FDA’s recent order to pull the medical device off the market. For now, the FDA is allowing the use of other surgical mesh products.
There are a few different varieties of mesh, each manufactured differently. These include:
- Animal-derived mesh
- Knitted synthetic mesh
- Non-knitted sheet synthetic mesh
Synthetic meshes of either texture are not absorbable by the body and remain in the body indefinitely. Animal-derived meshes, on the other hand, are absorbable. The surgical mesh used for transvaginal repair of POP is synthetic. It is a permanent implant intended to strengthen the weakened vaginal wall.
The FDA’s order for the withdrawal of these surgical mesh products should protect many women from the horrifying and painful complications of transvaginal surgical mesh. Approximately one in eight women will require surgical repair for POP, many of whom have had the transvaginal surgical mesh procedure.
What is Pelvic Organ Prolapse (POP)?
Pelvic Organ Prolapse (POP) occurs when the pelvic organs no longer have the support of the tissue and muscles of the pelvic floor. This causes the organs to drop, or prolapse, from their original position. The pelvic organ most often involved in POP is the bladder, but any pelvic organ may prolapse, including:
- Vagina
- Cervix
- Uterus
- Urethra
- Rectum
POP is caused by stretched or torn pelvic floor muscles. This is most often the result of:
- Labor
- Childbirth
- Genetic predisposition
- Connective tissue disorder
- Obesity
- Frequent constipation
POP is a very common condition among women, though not all women who have it show symptoms. Those who have it and have symptoms often experience:
- Pelvic discomfort and pain
- Pelvic pressure
- Bulge of tissue or organs protruding to or past the vaginal opening
- Urinary incontinence (leakage of urine)
- Sexual discomfort
There are non-surgical treatment options for POP, but many women have opted for the promise of a permanent treatment through the use of transvaginal mesh installation. Women who have had transvaginal POP repair with surgical mesh need routine check-ups for the rest of their lives to monitor the function of the device.
Unfortunately, many of the women who choose surgery experience complications and injuries caused by the surgical mesh. The FDA reports a significant increase in the number of reports of adverse events associated with the mesh in the last few years.
Surgical Mesh Injuries
The FDA halted the sale of surgical mesh for transvaginal POP repair as the final step in a long line of escalating safety warnings because manufacturers could not satisfactorily prove the risks of their product are worth the benefits offered. The FDA has warned for a long time that these surgical mesh products are risky. Among those warnings include:
- April 2019: The FDA ordered the immediate cessation of sale and distribution of these surgical mesh products.
- February 2019: The FDA convened a committee of gynecological experts to provide medical perspective on the effectiveness, safety, and risks of these devices.
- July 2018: The companies who failed to meet the requirements for Class III devices had to remove their mesh products from the medical device markets, further reducing the number of manufacturers making this type of mesh.
- January 2016: The FDA reclassified surgical mesh for transvaginal repair of POP as Class III, the highest risk class for devices.
- September 2011: After a public meeting with the Obstetrics and Gynecology Devices Panel about the risks of transvaginal surgical mesh, the FDA ordered 34 companies to perform post-market surveillance studies, a.k.a 522 Orders. In response, most manufacturers stopped selling the devices.
- July 2011: The FDA issued its first warning about surgical mesh for transvaginal repair of POP after receiving more than 1,500 reports of injuries caused by the device.
The overall problem with this surgical mesh product is that its use caused a host of complications and injuries, without measurably improving the lives of the women who received it. The complications women have suffered from transvaginal mesh include:
- Mesh erosion
- Pelvic pain
- Infection
- Bleeding
- Pain during sex
- Organ perforation
- Urinary problems
Many of these conditions require costly additional treatments including additional surgery. These side effects also have a tremendous emotional impact on patients and their loved ones.
What to Do if You Have Had a Transvaginal POP Repair with Surgical Mesh
If you have been treated with surgical mesh for POP, it is important that you take steps to protect your health. Continue to get annual routine check-ups if you have had this surgical repair. Your physician will want to monitor your device’s function closely. Unless your device starts to cause complications, there is no reason to take further medical action.
Seek medical attention right away if you start to experience symptoms like:
- Persistent vaginal bleeding
- Persistent vaginal discharge
- Pelvic or groin pain
- Pain during sex
These are the initial symptoms that the surgical mesh has begun to malfunction. Seek medical care right away to determine if pelvic mesh is the cause for your symptoms. If so, your next step should be contacting a medical device attorney.
Surgical Mesh Attorneys with Proven Results
The attorneys at Drug and Device Watch have handled many surgical mesh cases for women whose surgeries caused them pain and suffering instead of healing. The evidence that surgical mesh is not worth the risk finally became overwhelming. Sadly, for thousands of women, the damage has already been done.
If you have received surgical mesh to treat POP and have experienced complications, adverse side effects, or required surgery or additional treatment, you may be entitled to compensation. Contact Drug and Device Watch to find out more about your legal rights and your options. Call 1-888-458-6825, or complete our online form, to discuss your options.
Sources:
- https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/UCM636114.htm?utm_campaign=04162019_PR_FDA%20takes%20action%20on%20tranvaginal%20mesh%20for%20POP%20repair&utm_medium=email&utm_source=Eloqua
- https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/UroGynSurgicalMesh/default.htm
- https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/UroGynSurgicalMesh/ucm262299.htm
- https://www.mayoclinic.org/diseases-conditions/pelvic-organ-prolapse/in-depth/transvaginal-mesh-complications/art-20110300