Pharmaceutical company Pfizer has issued a medication recall for two lots of eletriptan hydrobromide because of potential contamination with bacteria. Eletriptan hydrobromide sells under the brand name Relpax. This recall is for the 40mg tablets of the drug. Does the recall of this popular medication affect you? Who is at...
Is Your Electronic Cigarette a Dangerous Product?
Are electronic cigarettes the next major dangerous product posing risk to consumers? Recent reports of illness, hospitalization, and even death linked to “vaping” certainly suggests that consumers are at risk. Vaping is the act of using an electronic cigarette (e-cigarette) to inhale vapor from liquid pods in the device. So...
Do Electronic Cigarettes Cause Neurological Side Effects?
The U.S. Food and Drug Administration (FDA) has received more than 100 reports of seizures and other neurological side effects in connection with the use of electronic cigarettes, or e-cigarettes. Despite seizures being a known complication of nicotine toxicity, the federal agency is not ready to officially link vaping to...
Doctors Recommend Treatment that Doesn’t Meet with FDA Approval
New information surfaces about drug safety and the risk of biologic medications administered outside of FDA approval. A group of prominent dermatologists issued a set of national guidelines for treating psoriasis in 2011. Nothing unusual, except that their recommendations do not meet with FDA approval. Furthermore, many of the doctors...
FDA Recalls Textured Breast Implants over Cancer Concerns
In July, 2019, the U.S. Food and Drug Administration (FDA) requested that Pharmaceutical manufacturer, Allergan, recall certain textured breast implants. FDA recalls like this one highlight ongoing concerns about breast implants and resulting illnesses. It also continues the discussion about whether a warning or a medical device recall is more...
Bayer Issues a Recall of Mislabeled Biologic Medications
Pharmaceutical giant Bayer is voluntarily recalling nearly one thousand vials of a biological drug used to treat patients with hemophilia A. The U.S. Food and Drug Administration (FDA) published the recall in July, 2019 explaining the danger to patients who rely on the drug. Medication recalls relating to mislabeled drugs...
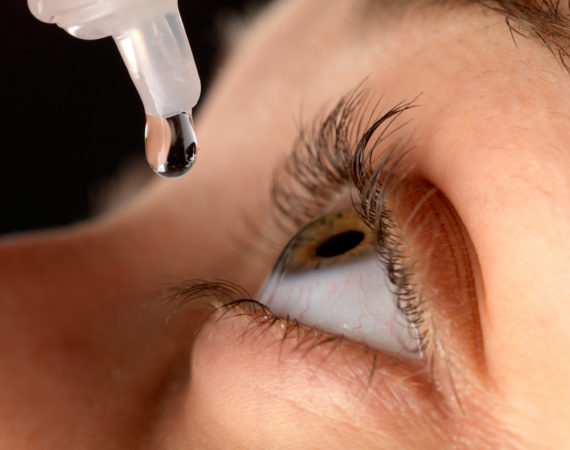
Massive Dry Eye Medication Recall Causes Shortages
One after another, eye drop manufacturers have announced a medication recall for drops used to treat dry eye. It started with a recall of generic brands and quickly spread to the name brands. At this point, experts are calling on manufacturers who have NOT recalled their eye drops to speak...