Medical device manufacturer, Roche Diagnostics, has received emergency use approval (EUA) from the U.S. Food and Drug Administration (FDA) for their Cobas SARS-CoV-2 coronavirus test. The test detects the virus COVID-19 through nasopharyngeal or oropharyngeal swab samples. The test can be used in patients who meet clinical or epidemiological testing...
Drug Manufacturers Developing Vaccines and Treatments for Coronavirus
The number of coronavirus (COVID-19) cases in the United States continues to rise. As more communities suffer infection, quarantines and general panic, researchers continue to learn whatever they can about how the virus spreads, who is most at risk and how the virus could be prevented. Drug manufacturers are among...
FDA Urged to Recall Metformin after Third Party Testing Shows NDMA in 16 Batches
Pharmaceutical companies are under more pressure than ever due to concerns about contamination and the presence of unsafe ingredients. Readers will no doubt recall notable recent medication recalls like Zantac (ranitidine), Valsartan and Belviq. Now, another popular drug, Metformin, is joining those ranks after third party tests found the probable...
Weight Loss Drug Belviq Recall: Drug Linked to ‘Increased Occurrence of Cancer’
Popular weight loss drug Belviq is being recalled after a safety clinical trial showed an “increased occurrence of cancer.” Following the trial, the U.S. Food and Drug Administration (FDA) requested a Belviq recall from Eisai Co., the manufacturer. Eisai acceded to the FDA’s request, though the company claims that their...
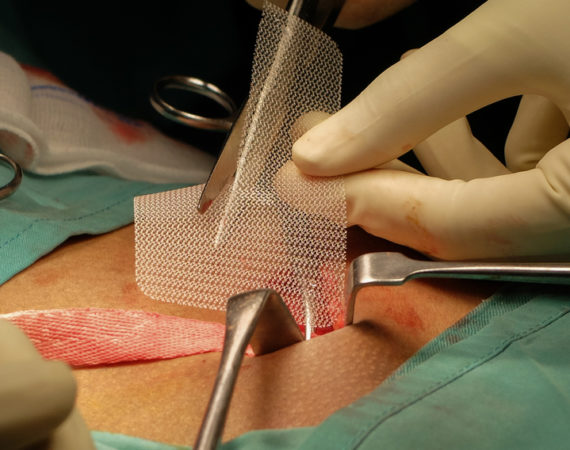
Trial Dates Set for Bard Hernia Mesh Cases
After years of moving through the legal process, the first trial dates are set for consolidated Bard hernia mesh cases. In January 2020, plaintiffs and defendants each selected the cases they wanted to proceed to trial. The court will choose three cases to proceed to bellwether trials. Once case-specific motions...
FDA Announces Surgical Gown Recall for more than 9 Million Units
The U.S. Food and Drug Administration (FDA) is warning healthcare facilities to stop using certain surgical gowns made in China due to the possibility that they are not sterile. The recall issuer is medical device manufacturer Cardinal Health, but the gowns were made in China at a contractor facility. The...
Cybersecurity Warnings Issued for GE Healthcare Medical Devices
The U.S. Food and Drug Administration (FDA) has issued a device safety alert for GE Healthcare medical devices. The safety alert relates to GE Healthcare Clinical Information Central Stations and Telemetry Servers. The FDA says that cybersecurity threats could risk the health of the patients that these medical devices monitor...
More Antacid Recalls Issued due to Trace Elements of NDMA
On the heels of notable recent medication recalls like Zantac, the U.S. Food and Drug Administration (FDA) is recalling even more medications due to possible trace elements of NDMA. NDMA (Nitrosodimethylamine) is a probable human carcinogen, meaning it may cause cancer. The presence of NDMA in Zantac has made waves...
Investigation Continues in Lung Injury Outbreak Related to E-Cigarettes
The investigation into the ongoing lung injury outbreak continues, with the focus remaining on e-cigarette use as the culprit. The Centers for Disease Control and Prevention (CDC), U.S. Food and Drug Administration (FDA) and various local and state health departments are joining forces to continue the investigation. The lung injury...
FDA Recall Alert: Antidepressant Mirtazapine Affected by Dangerous Labeling Error
The U.S. Food and Drug Administration (FDA) has announced that Aurobindo Pharma USA, Inc. is voluntarily recalling a single lot of antidepressant medication, Mirtazapine. According to the FDA notice, the recall is due to a dangerous labeling error. Patients could take a higher dose of the antidepressant than what is...
Mavidon is Recalling All Medical Devices due to Burkholderia Cepacia Contamination
Mavidon – a large manufacturer of medical devices – is recalling all of their products worldwide due to possible contamination with Burkholderia cepacia. This worldwide medical device recall is making waves in the healthcare industry. Here is what we currently know about the recall. Worldwide Recall of Mavidon Medical Devices...